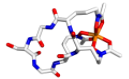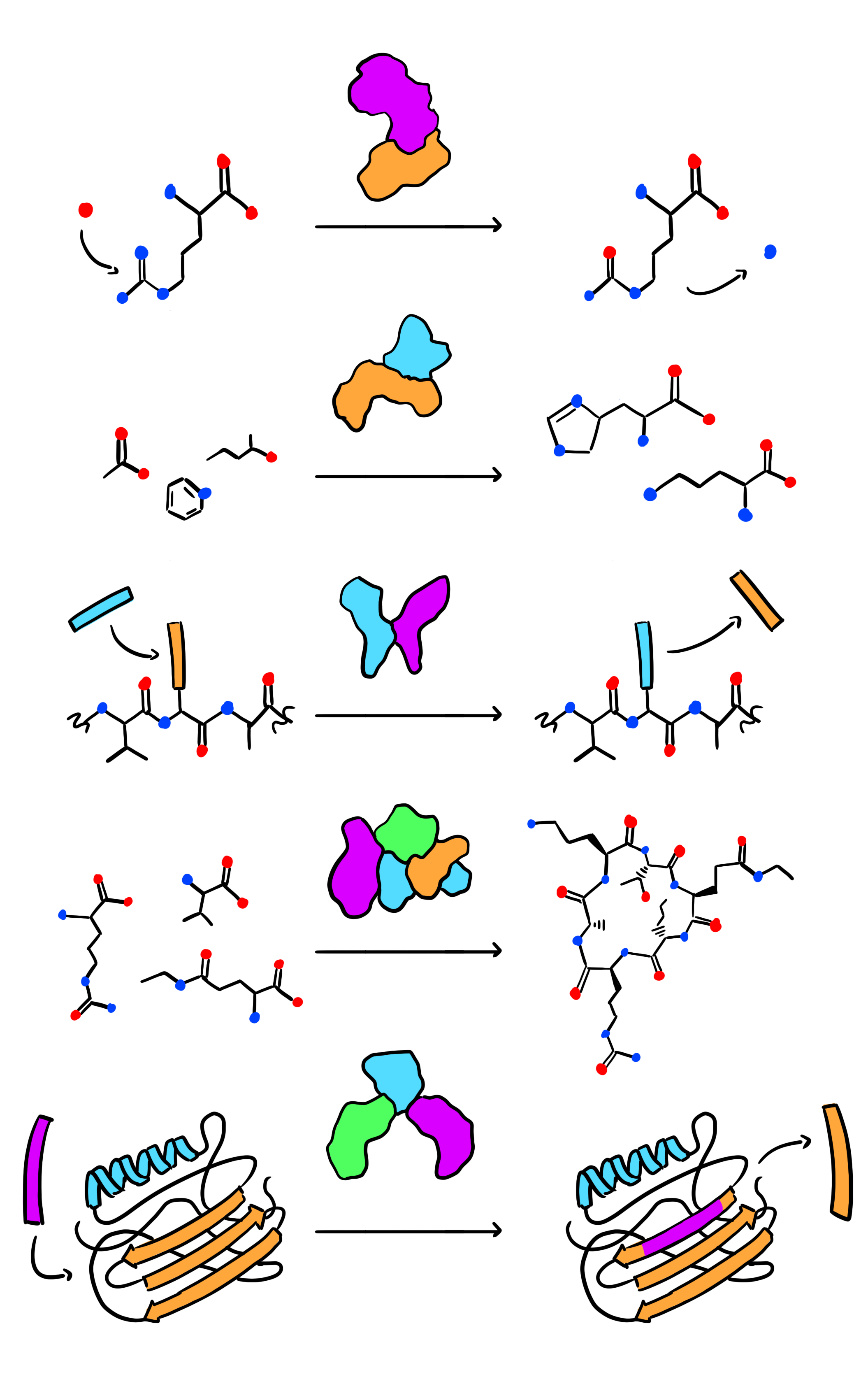
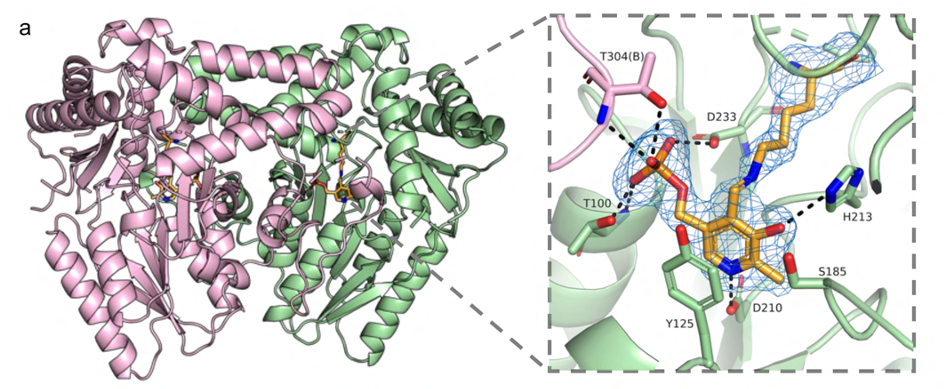
Enzymology and biocatalysis of PLP-dependent enzymes
We focus on discovering and understanding new PLP-dependent enzymes from natural product biosynthetic pathways and developing them into biocatalysts for non-canonical amino acid biosynthesis. Besides, we are also interested in deorphanizing functionally unknown human PLP-dependent enzymes and developing chemical therapeutics.
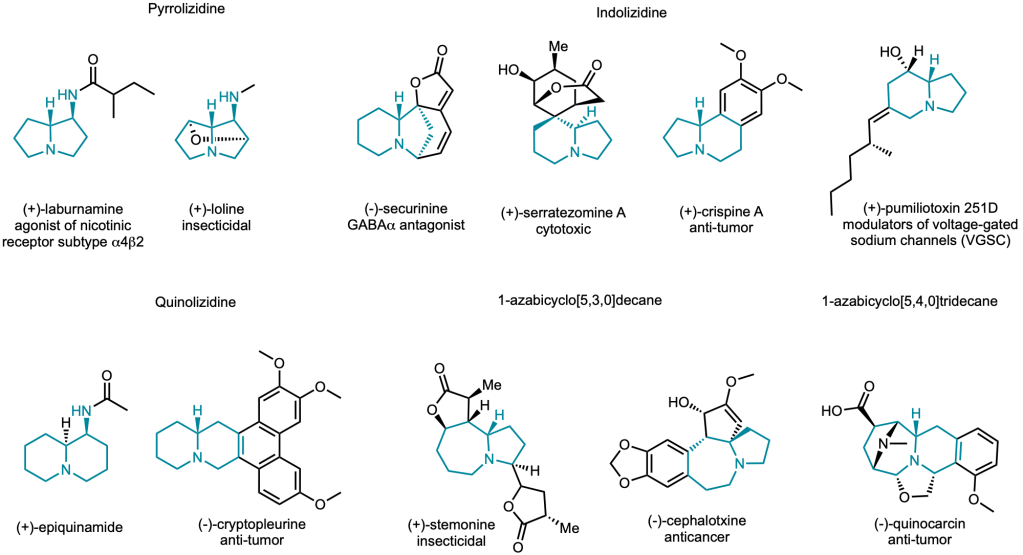
Biosynthesis of bioactive alkaloids
We are interested in elucidating the biosynthesis of alkaloid natural products and developing synthetic biology platform to build artificial biosynthetic pathways to access bioactive and structurally complex alkaloid. We aim to expand the chemical diversity and ultimately build unnatural bioactive alkaloids as therapeutic agents.